We conducted a double digest restriction associated DNA study on Mimulus gutattus. The first step was collecting samples, which was done on two field trips (Lab 02 and Lab 05)
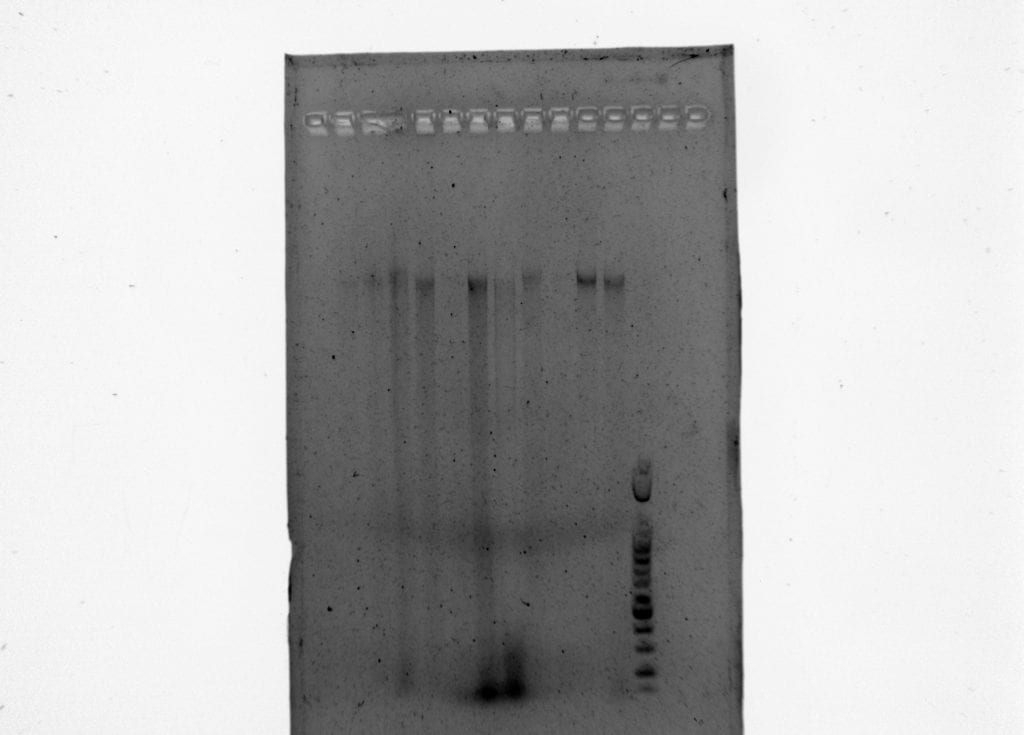
Next, we extracted DNA from collected samples as well as previously collected samples by Our Boy Alec. After, we double digested our DNA using two restriction enzymes, cutting the DNA into many pieces. Then, we ligated unique DNA barcodes onto each individual. Our next step was using PCR to add a second unique barcode and to test if our library construction was successful. The library construction was successful. After the test PCR, a larger reaction was conducted that was identical to the aforementioned process. This was the last step performed as a class and in a perfect world, the next steps would start with size selection. Size selection selects DNA of specific sizes (duh), but specifically we would be targeting 400-600 bp fragments. This is the typical size for Illumina technology based studies. We could use PippenPrep to do this, of which we have housed in the Suni Lab (#Suni!!!) Alternatively, we could run out the PCR on a gel extraction to cut them based on a ladder. Lastly, you could use metallic beads to isolate the DNA.
After size selection, DNA size samples would be normalized–bringing all of them to approximately the same concentration. The final step after that would be to combine all the size selected and normalized PCR products into one vessel. Then these samples wold be run on the in-house Illumina sequencer. Sequencing would take ~16 hours and if successful, would generate tens of millions of reads. These data would be run through a bioinformatics pipeline (looks at Dr. Zimmerman). Ultimately we would align these data with the published M. guttatus genome and call SNPs. We would use these SNPs to infer population differentiation using a metric like FST and assess population genetic diversity looking at things like the number of alleles, allelic diversity, etc.
Based on what we know about M. guttatus, geographically diverged samples will probably have larger population differentiation than those who are geographically closer to each other.