Date: 11/1/17
Location: G04
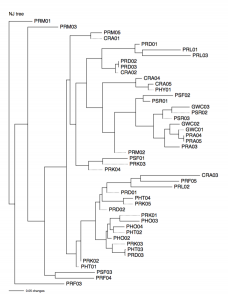
Test Gel
We added 1ul of dye with a micropipetters to our ISSR Plant DNA tubes from the previous class. We then got 10ul of this and placed each sample into a gel for about 1h 45min at 60v. the gel was 1.3x agarose. The primer that we had was OMAR.
ISSR PCR
Dilutions
We got our plant DNA samples from previous class and made dilutions with it. The samples that I had were PSR 01-05. I micropipetted 10ul of the DNA sample into a 1.5ml microfuge tube and added 90ul of H20 so it was a 1:10 dilution.
PCR Setup
For each Primer, Omar and 1788, we got 2 0.2ml 8-tube strips in each I put each of the Plant DNA sample dilutions. I had 6 tubes, one for each of my 5 samples and 1 extra for the negative. My samples were
PSR 01
PSR 02
PSR 03
PSR 04
PSR 05
With a micropipetter, I put 1ul of each sample into each tube for each of my samples.
For each of our PRIMERS (either Omar or 1788), we made a master mix. The contents of those master mixes were:
ddH20: 15.4ul
10xbuffer: 2.00ul
BSA: 1.0ul
dNTPs: 2.0ul
Primer : 0.20ul
Taq: 0.04ul
Template: 1.0ul
Total: 20ul
We then placed 19ul of master mix into each of the tubes in which they were placed in a PCR machine.